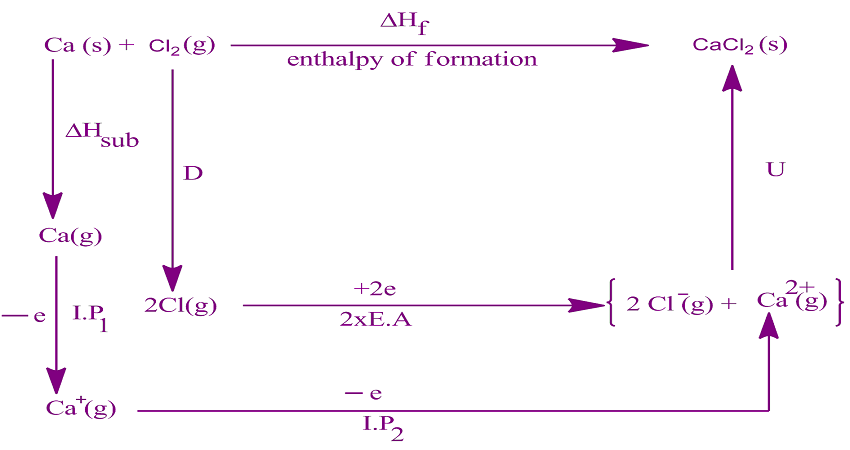
M 2+O 2- materials have high lattice energies vs. sodium chloride due to greater charge/radius for both cation and anion The following table presents a list of lattice energies for some common compounds as well as their structure type.ĭifference vs. It has been shown that neglect of polarization led to a 15% difference between theory and experiment in the case of FeS 2, whereas including it reduced the error to 2%. As an example, one may consider the case of iron-pyrite FeS 2. In these cases the polarization energy E pol associated with ions on polar lattice sites may be included in the Born–Haber cycle. Effect of polarization įor certain ionic compounds, the calculation of the lattice energy requires the explicit inclusion of polarization effects. The bond radii are similar but the charge numbers are not, with BaO having charge numbers of (+2,−2) and NaCl having (+1,−1) the Born–Landé equation predicts that the difference in charge numbers is the principal reason for the large difference in lattice energies.Ĭlosely related to this widely used formula is the Kapustinskii equation, which can be used as a simpler way of estimating lattice energies where high precision is not required.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
